Science: Targeting Angiogenic Metabolism in Disease
By Dr. Xuri Li from Zhongshan Ophthalmic Center, Sun Yat-Sen University and Dr. Peter Carmeliet from VIB-KU-Leuven
Edited by: Liu Nian
On March 23, 2018, an article titled “Targeting Angiogenic Metabolism in Disease” was published by the journal of Science, in which Dr. Xuri Li from the Zhongshan Ophthalmic Center (ZOC), Sun Yat-Sen University (SYSU) and Dr. Peter Carmeliet from VIB-KU Leuven in Belgium discuss the importance and promise of an immerging new field of vascular research, endothelial cell (EC) metabolism.
In this article, Dr. Li and Dr. Carmeliet elucidate the role and mechanism of vascular EC metabolism in relationship to numerous human diseases, such as ocular neovascular diseases and tumors, and propose that manipulating EC metabolism may offer new therapeutic opportunities for the treatment of various diseases.

Dr. Myriam Baes, Dr. Peter Carmeliet, Dr. Yizhi Liu, Dr. Luc Schoonjans,Dr. Xuri Li and Dr. Weisi Lu (from left to right) at ZOC, SYSU discussing collaborative work
The vascular system is one of the most important organs in the human body. It supplies oxygen and nutrients to all tissues and organs, and maintains their normal morphologies and functions. Dysregulation of the vascular system leads to vascular malfunction, damage or neovascularization, which subsequently trigger or accelerate various diseases. For more than 40 years, the research and development of antiangiogenic drugs have mainly focused on angiogenic factors, for example, vascular endothelial growth factor (VEGF). However, many challenges exist, including poor or no response and drug resistance, thus warranting a fundamentally different approach.
In recent years, EC metabolism has been shown to play critical roles in angiogenesis. Under pathological conditions, the blood vessels switch from a quiescent state to an activated one. Upon the actions of angiogenic factors, vascular ECs proliferate, migrate and differentiate into stalk and tip cells to form new blood vessels (left part in the below figure). During this process, vascular cells need a large amount of energy through high levels of metabolism. Hence, EC metabolism is crucial for vascular growth.
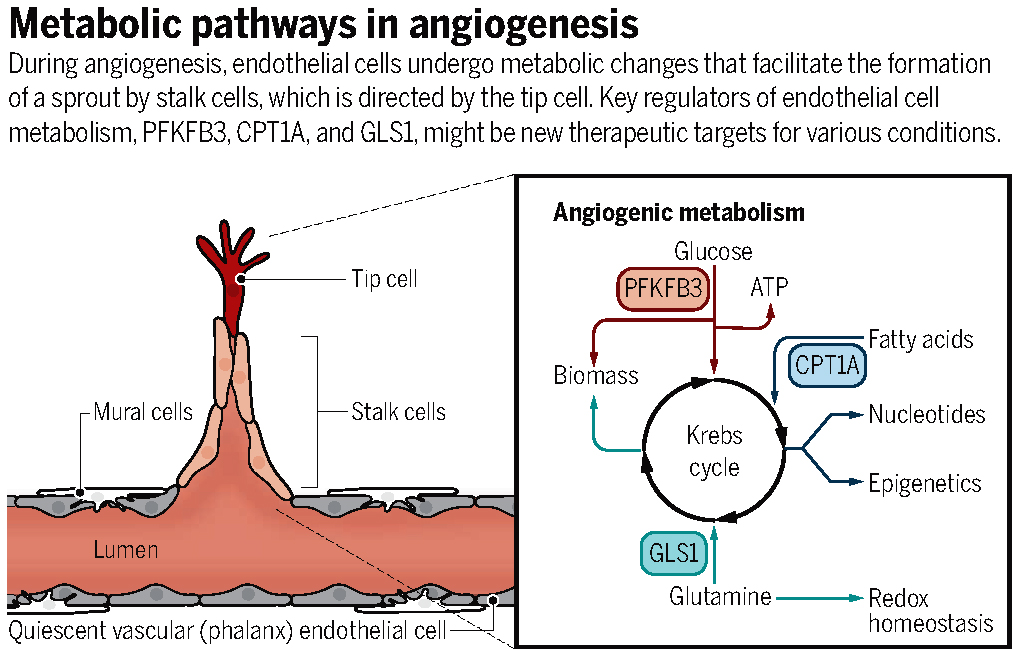
Several metabolic pathways in ECs have been characterized recently (right part in the above figure), thus yielding new therapeutic targets for antiangiogenic therapy. These pathways include EC glycolysis regulated by PFKFB3 and FGF2, EC fatty acid oxidation mediated by CPT1A, glutamine metabolism regulated by GLS1, asparagine metabolism modulated by ASNS, etc. The field of EC metabolism research is still in its infancy. However, future studies on the function and mechanism of EC metabolism hold high promise for more novel insight into vascular diseases and possibilities for better therapies.
Dr. Carmeliet is an elected member of the German Academy Sciences Leopoldina, Dutch Royal Academy of Sciences, EU Academy of Sciences, and European Molecular Biology Organization. Dr. Carmeliet is a Professor at VIB-KU Leuven, which has ranked number one for many years in Europe in terms of innovation. KU Leuven was founded in 1425 and is one of the oldest and most prestigious universities in the world. Dr. Carmeliet has published more than 646 publications (94 IF>20, 243 IF>10, h-index 126) till March 2018, and is ranked by Science Match as top eight most cited authors in General Biomedicine. Dr. Carmeliet is on the editorial board of Cancer Cell, was a consulting editor for Science, and has more than 72 patents. Based on Dr. Carmeliet’s research, several biocompanies have been founded and clinical trials have been launched.
Dr. Xuri Li is the Scientific Director of the State Key Laboratory of Ophthalmology at the Zhongshan Ophthalmic Center (ZOC), Sun Yat-Sen University in Guangzhou, China. Before joining ZOC, Dr. Li served as a Principal Investigator at the National Institutes of Health, National Eye Institute in the US during 2005-2012. SKLO is the only one of its kind in China and is supported by the central Chinese government. ZOC is the all-time number one eye hospital in China. SKLO and ZOC constitute the largest eye care and research center in the world. SYSU is the 5th top-ranked university in China and the 2nd in terms of campus area.
References
1 Li, X. & Carmeliet, P. Targeting angiogenic metabolism in disease. Science 359, 1335-1336, (2018).
2 Carmeliet, P., Li, X., Treps, L., Conradi, L. C. & Loges, S. RAISEing VEGF-D's importance as predictive biomarker for ramucirumab in metastatic colorectal cancer patients. Ann Oncol, Jan. 19, (2018).
3 Wong, B. W. et al. The role of fatty acid beta-oxidation in lymphangiogenesis. Nature 542, 49-54, (2017).
4 Cantelmo, A. R. et al. Inhibition of the Glycolytic Activator PFKFB3 in Endothelium Induces Tumor Vessel Normalization, Impairs Metastasis, and Improves Chemotherapy. Cancer Cell 30, 968-985, (2016).
5 Quaegebeur, A. et al. Deletion or Inhibition of the Oxygen Sensor PHD1 Protects against Ischemic Stroke via Reprogramming of Neuronal Metabolism. Cell Metabolism 23, 280-291, (2016).
6 Rafii, S. & Carmeliet, P. VEGF-B Improves Metabolic Health through Vascular Pruning of Fat. Cell Metabolism 23, 571-573, (2016).
7 Schoors, S. et al. Fatty acid carbon is essential for dNTP synthesis in ECs. Nature 520, 192-197, (2015).
8 Ghesquiere, B., Wong, B. W., Kuchnio, A. & Carmeliet, P. Metabolism of stromal and immune cells in health and disease. Nature 511, 167-176, (2014).
9 Schoors, S. et al. Partial and transient reduction of glycolysis by PFKFB3 blockade reduces pathological angiogenesis. Cell Metabolism 19, 37-48, (2014).
10 De Bock, K., Georgiadou, M. & Carmeliet, P. Role of EC Metabolism in Vessel Sprouting. Cell Metabolism, (2013).
11 De Bock, K. et al. Role of PFKFB3-Driven Glycolysis in Vessel Sprouting. Cell 154, 651-663, (2013).
